|
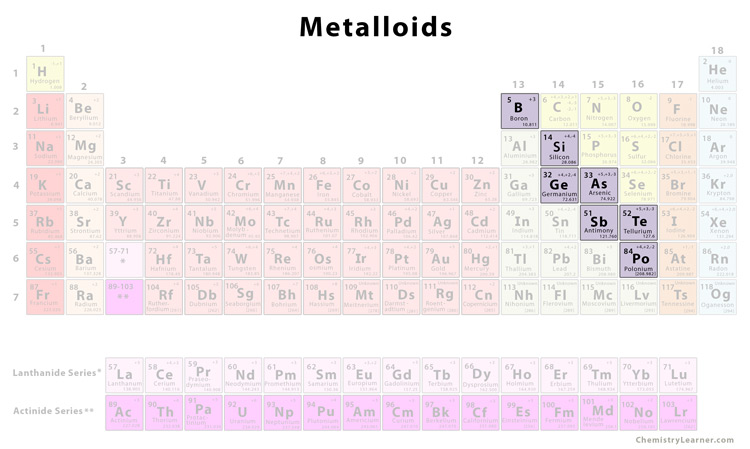
If you’re reading this article on any sort of electronic device - cell phone, laptop, computer or tablet- then you’re utilizing silicon. It earned this moniker because it is gray and metallic when in a crystalline form, resembling the stone it borrows its name from. It’s the second most abundant element in the Earth’s crust and gets its name from silex, the Latin word for Flint. Silicon is easily one of the most useful elements on the planet. What are some uses of metalloids in our daily life? Where might you find these elements during your daily activities? Most of them are more common than you might think. This trait, of only conducting electricity under certain circumstances, is known as semiconductivity. One of the most coveted properties of metalloids is the fact that under certain conditions, they can act as conductors. Boron, for example, will act like a nonmetal when interacting with the volatile element sodium but will behave like a metal element when interacting with fluorine. Whether or not these elements present metallic properties depends on the substances with which they’re interacting. They can, and generally do, form alloys with the metals around them, making them stronger and giving them metallic properties. They look like brittle metals, but their chemical behavior is distinctly non-metallic. The only element more abundant is oxygen.Īnother thing that is strange about the semimetals on the periodic table is that they don’t appear on the periodic table in a straight vertical or horizontal line – their group line is diagonal.Įlements belonging to the semimetals group can be strange. 
Silicon is the second most abundant element found in the Earth’s crust. The elements ‘officially’ included in the semimetals group are: Even scientists have trouble answering the question ‘what are metalloids?’ For most scientists, it’s a judgment call. There’s no definitive answer as to which group these elements belong. Metalloids, also known as semimetals, fit between the metals and nonmetals on the periodic table, but what are metalloids, really? They are tricky to define because they have what is known as intermediate properties, meaning they can present properties that could belong to either the metals or nonmetals group. What are metalloids and where might you use them during your everyday activities? Today, we’re going to take a closer look at some elements that you might not encounter - or want to encounter - during your daily activities: the semimetals. We encounter a variety of different elements every single day, from the fluoride in your toothpaste to the sodium and chlorine in your table salt, but some of them are more common than others. When you buy through links on our site, we may earn an affiliate commision. Antimony gets its name from the Greek words "anti monos" meaning "not alone.Revolutionized is reader-supported.Antimony has been known about since ancient times and was used as a cosmetic by the Ancient Egyptians.Tellurium gets its name from the Latin word "tellus" which means "earth.". 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
